einstein (São Paulo). 01/Oct/2015;13(4):555-9.
Bone health in cerebral palsy and introduction of a novel therapy
DOI: 10.1590/S1679-45082015AO3321
ABSTRACT
Objective
To assess the bone health status of children with cerebral palsy and the therapeutic effect of denosumab in a subgroup of children with cerebral palsy and decreased bone mass.
Methods
Children with cerebral palsy were evaluated according to their motor disability score (classification system gross motor functions III to V), bone density and bone turnover markers. Dual X-ray energy absorption was used to measure the lumbar spine, and total body, except the head. Thereafter a group of children with cerebral palsy and osteoporosis was treated with denosumab, a fully human monoclonal antibody. Bone turnover markers were measured before and three months after treatment.
Results
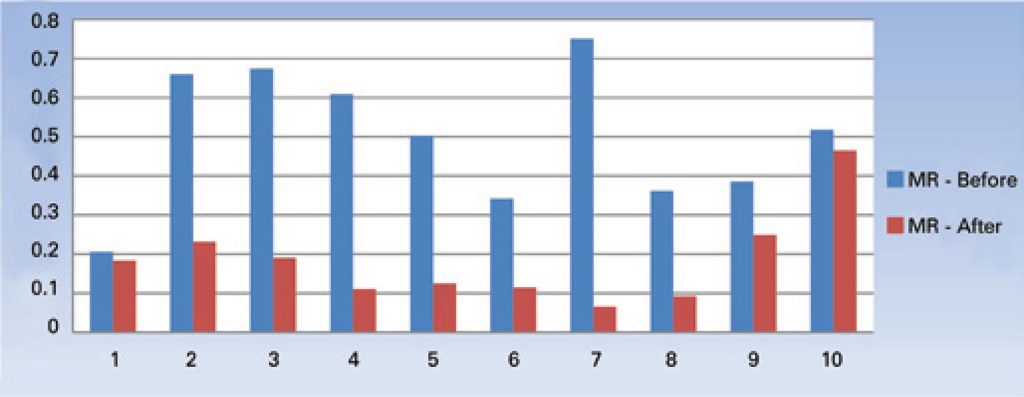
Reduction in bone mineral density was observed, particularly in children with greater impairment evaluated by the motor score. Decreased bone turnover markers were found in a selected group of children three months after exposure to denosumab.
Conclusion
Bone loss was present in children with significant impairment of motor function, as well as decreased serum levels of bone resorption markers with new forms.
533