einstein (São Paulo). 31/Jan/2014;11(4):435-8.
Paullinia cupana for control of hot flashes in breast cancer patients: a pilot study
DOI: 10.1590/S1679-45082013000400005
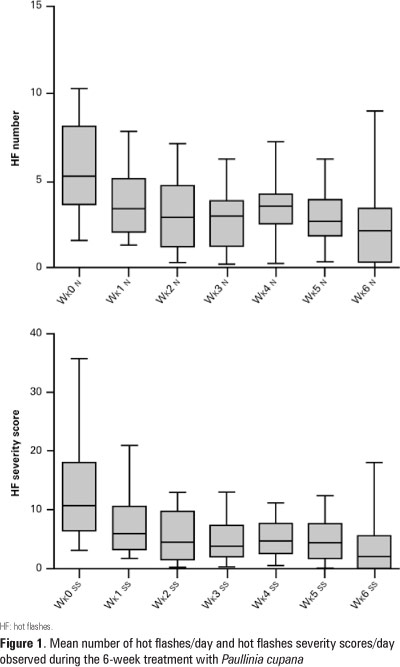
OBJECTIVE: To evaluated whether Paullinia cupana decrease number and severity of hot flashes in breast cancer survivors. METHODS: This was a prospective phase II pilot study. We studied female breast cancer survivors who had completed the cancer treatment 3 months previously and who were experiencing at least 14 hot flashes per week. At least 9 of the 15 patients were required to have a decrease of at least 50% in hot flash severity score in keeping with the Simon Design. Patients received 50mg of dry extract of Paullinia cupana orally twice a day for 6 weeks. We assessed both frequency and severity of hot flashes. RESULTS: A total of 18 patients started the Paullinia cupana treatment, and 15 completed the study. Three patients left the study immediately after starting the treatment because of personal difficulties in participation or noncompliance. Of the 15 patients who completed the study 10 had a decrease of more than 50% in hot flash severity scores. During the 6 weeks of treatment, statistically significant decreases were seen in both numbers of hot flashes (p=0.0009) and severity scores (p<0.0001). Paullinia cupana was well tolerated, and there were no instances of discontinuation because of toxicity. CONCLUSIONS: Paullinia cupana appears promising for controlling hot flashes. More extensive studies seem warranted.
Keywords: Breast neoplasms; Hot flashes; Paullinia pinnata; Guarana (Homeopathy)
228