einstein (São Paulo). 01/Jul/2019;17(4):eRC4599.
Dupilumab in the treatment of severe atopic dermatitis refractory to systemic immunosuppression: case report
DOI: 10.31744/einstein_journal/2019RC4599
ABSTRACT
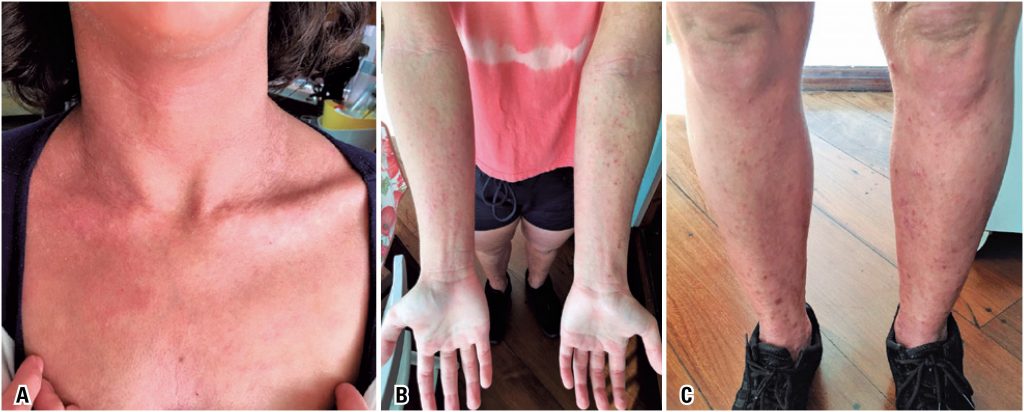
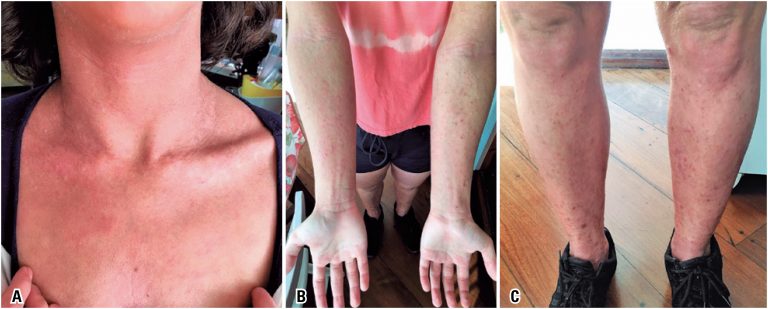
Case report of a patient with severe atopic dermatitis who showed a good response to dupilumab. She had already used two immunosuppressive agents, cyclosporine A and mycophenolate mofetil, for the treatment of atopic dermatitis with no proper control of the disease. She had also been taking all measures to control severe cases of the disease: bath and environmental controls, topical potent corticosteroids and emollients. She presented constant pruritus and skin lesions, frequent skin infections e poor quality of life. She also developed depression due to her disease. Recently, dupilumab, a new biological agent, was approved for the treatment of moderate/severe atopic dermatitis in many countries, including Brazil. Dupilumab is a monoclonal antibody with a common alpha chain of interleukin (IL) 4 and IL-13 receptors, two cytokines involved in the Th2 profile immune response that promote atopic inflammation. In a pioneer way in Brazil, the patient initiated the treatment with an attack dose of 600mg subcutaneous of dupilumab and 300mg subcutaneous every other week. Up to now, she has taken four applications, presenting a great improvement of the disease and her quality of life. There were no adverse effects, nor in the injection site nor of other kind. Patient and her family are very satisfied, and the medical team evaluates that the treatment is being well succeed. The case report described here subsidizes the use of dupilumab in the treatment of severe atopic dermatitis refractory to use of immunosuppressive agents.
7,124