einstein (São Paulo). 21/Nov/2018;16(4):eAO4279.
Percutaneous laser ablation of benign thyroid nodules: a one year follow-up study
DOI: 10.31744/einstein_journal/2018AO4279
ABSTRACT
Objective
To evaluate safety and effectiveness of nodule volume reduction and thyroid function after percutaneous laser ablation treatment in patients with benign nonfunctioning thyroid nodules.
Methods
Prospective single-center study, from January 2011 to October 2012, which evaluated 30 euthyroid and thyroid antibodies negative patients with benign solitary or dominant nodule with indication of treatment due to compressive symptoms and aesthetic disturbances. The clinical and laboratory (thyroid ultrasound, TSH, FT4, TG, TG-Ab, TPO-Ab and TRAb levels) evaluations were performed before the procedure, and periodically 1 week, 3 months and 6 months after. The ablation technique was performed under local anesthesia and sedation. In each treatment, one to three 21G spinal needle were inserted into the thyroid nodule. The laser fiber was positioned through the needle, which was then withdrawn 10mm to leave the tip in direct contact with the nodule tissue. Patients were treated with a ND: Yag-laser output power of 4W and 1,500 to 2,000J per fiber per treatment. The entire procedure was performed under US guidance.
Results
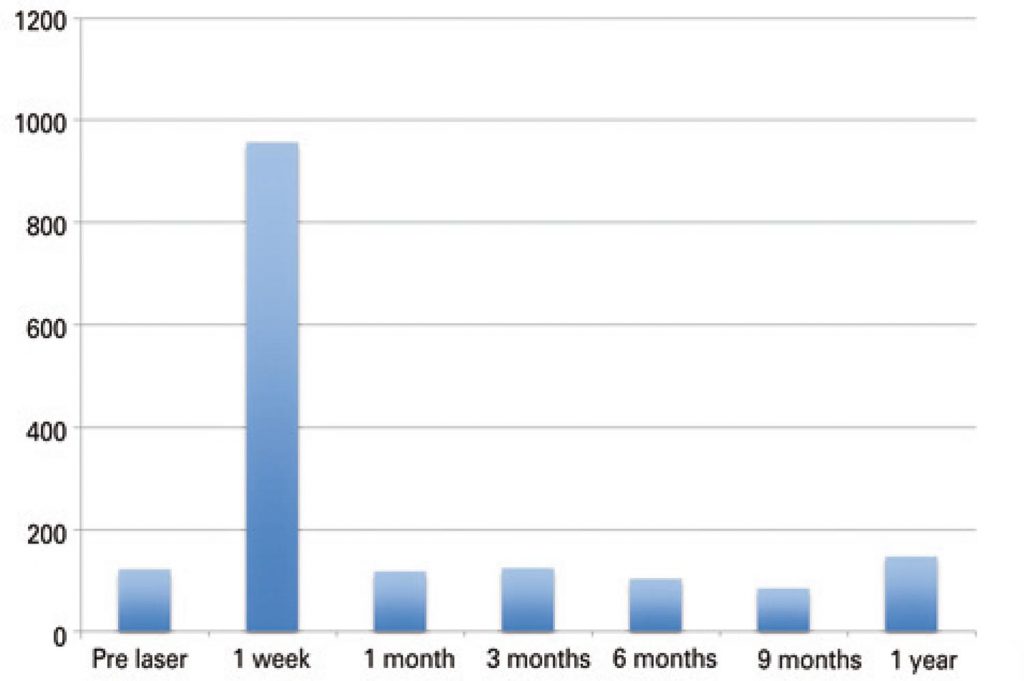
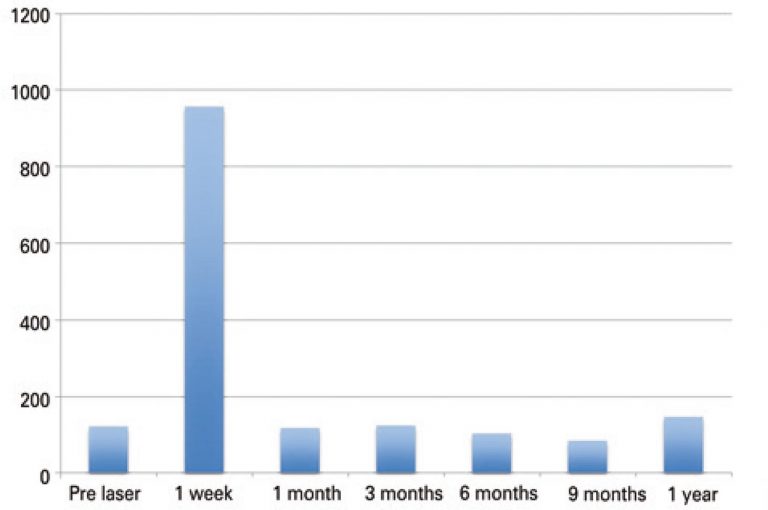
Thirty patients, with a total of 31 nodules submitted to laser ablation were evaluated. The median volumetric reduction of the nodule was approximately 60% after 12 months. No statistical significance was observed on thyroid function and antibodies levels. There was a peak on the level of thyroglobulin after the procedure due to tissue destruction (p<0.0001). No adverse effects were observed.
Conclusion
Percutaneous laser ablation is a promising outpatient minimally invasive treatment of benign thyroid nodule.
Keywords: Lasers; Ablation technique; Thyroid nodule; Brazil
1,540