einstein (São Paulo). 01/Oct/2011;9(4):436-41.
Nanostructured SBA-15 silica as an adjuvant in immunizations with hepatitis B vaccine
DOI: 10.1590/S1679-45082011AO2162
ABSTRACT
Objective:
To evaluate the applicability of SBA-15 silica as an adjuvant in immunizations with purified particles of the viral protein HBsAg, the main component of hepatitis B vaccine, Butang®, produced by Instituto Butantan.
Methods:
BALB/c mice orally or subcutaneously received 0.5 μg of HBsAg adsorbed/encapsulated to SBA-15 or adsorbed to Al(OH)3. To assess the secondary immune response, a subcutaneous booster was administered 30 days after the first immunization. Individual serum and fecal samples of each group were periodically collected for specific antibody titration by ELISA.
Results:
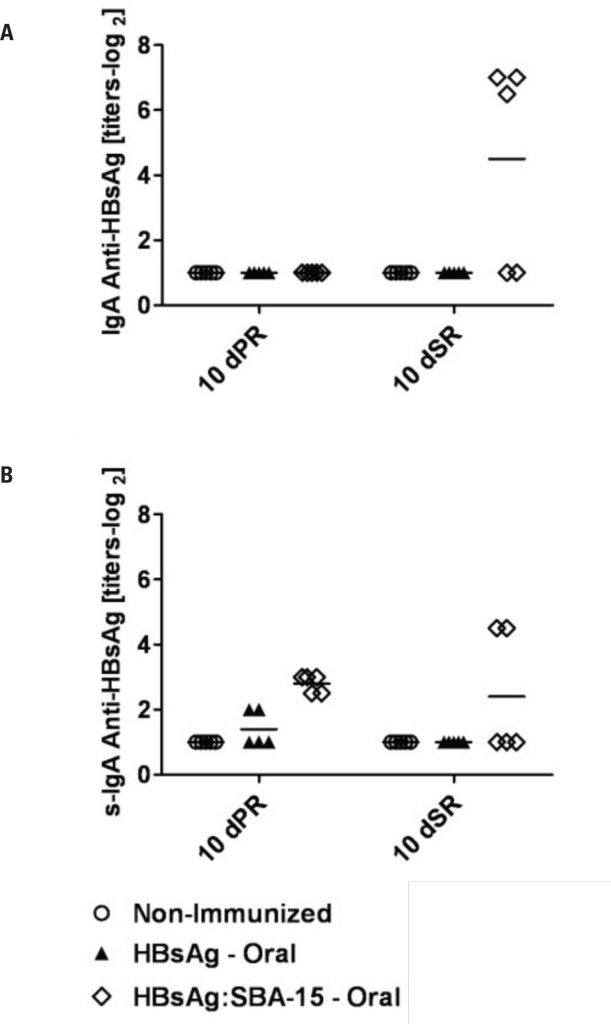
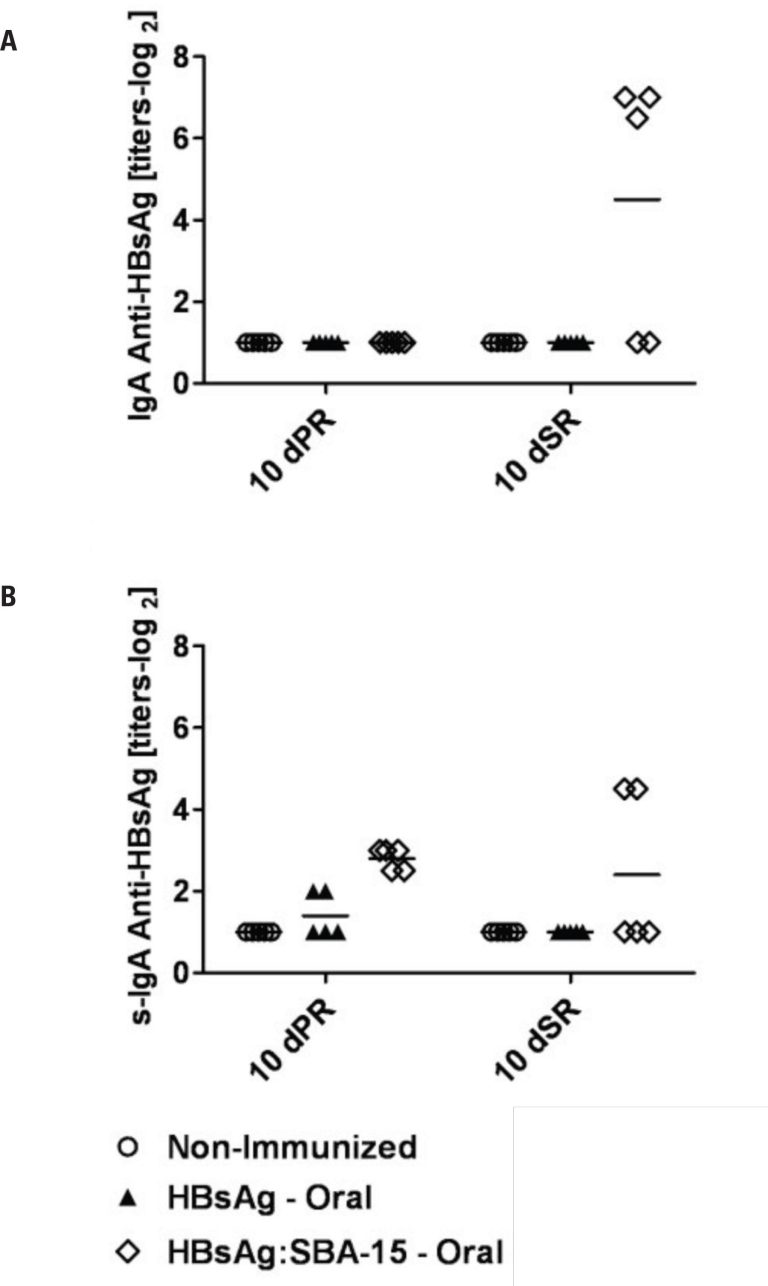
Analysis of secretory IgA showed that mice orally primed with HBsAg on SBA-15 had increased levels of specific antibodies in primary and secondary immune responses. Specific serum IgA and IgG titers in HBsAg:SBA-15-orally immunized mice reached higher levels after the booster, demonstrating the effectiveness of oral vaccination with the use of silica. All immunized groups showed higher IgG1 levels.
Conclusion:
Our results clearly indicate the promising use of SBA-15 as an adjuvant, especially in oral immunizations.
Keywords: Hepatitis B; Oral vaccination; Adjuvants; Immunological memory
224