einstein (São Paulo). 22/May/2019;17(3):eAO4521.
Drug interactions in maternal intensive care: prevalence, risk factors, and potential risk medications
DOI: 10.31744/einstein_journal/2019AO4521
ABSTRACT
Objective:
To characterize severe potential drug interactions in maternal intensive care, and to determine their frequency, risk factors and potential risk medications.
Methods:
An observational and longitudinal study conducted between December 2014 and December 2015 in a maternal intensive care unit. Clinical data were collected and severe potential drug interactions were identified on pregnant inpatients. The drug interactions were classified by type, prevalence and exposure rate. A multivariate logistic regression model was used to identify the severe potential drug interactions and the related drugs (p<0.05).
Results:
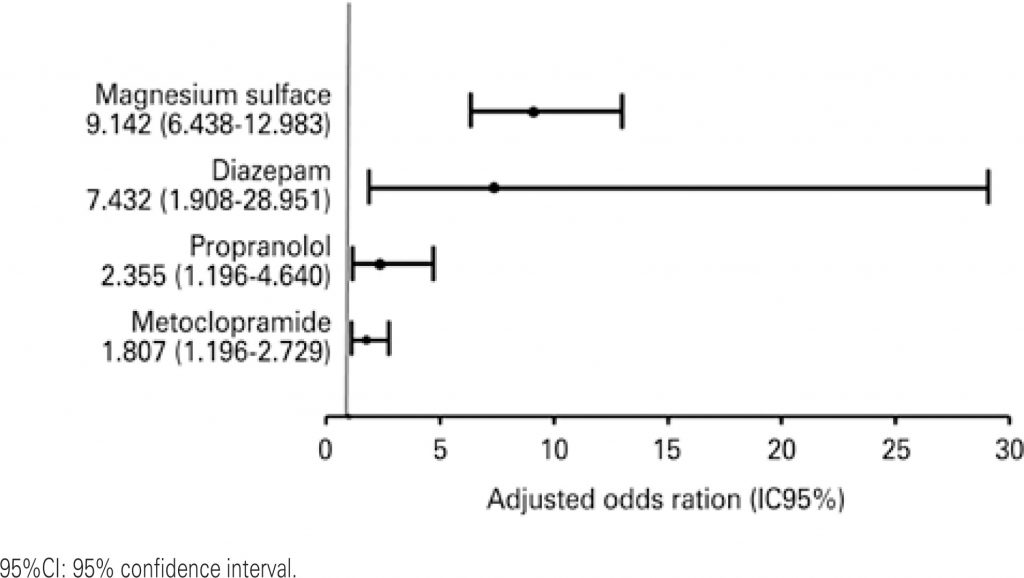
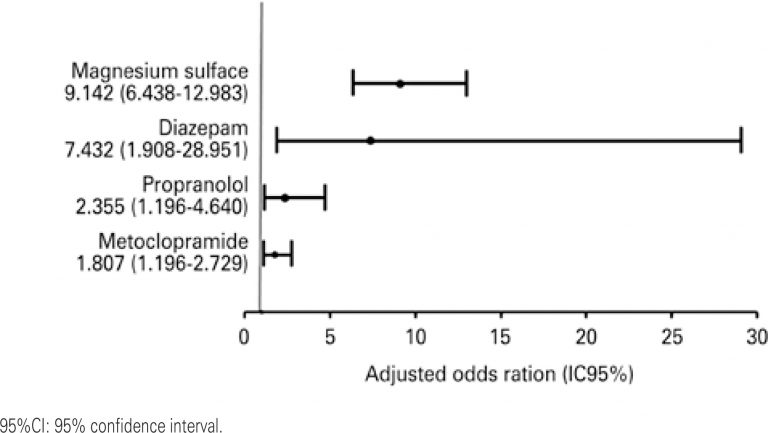
A total of 95.1% of patients were exposed to, at least, one potential drug interaction; in that, 91.7% 33.9% were related to, respectively, moderate and severe potential drug interactions. The patients were exposed, on average, on 69.2% of days they were in the intensive care unit. The main drugs involved in more severe drug interactions were magnesium sulfate, metoclopramide, propranolol and diazepam.
Conclusion:
The severe potential drug interactions were observed in almost all patients of the study, and, approximately one third of those interactions were related to greater severity and resulted in exposure during long hospital stay. The higher number of prescribed drugs and its previous use of medications at home increase the occurrence of severe potential drug interactions.
1,393