einstein (São Paulo). 01/Jul/2011;9(3):265-82.
Cochrane meta-analysis: teicoplanin versus vancomycin for proven or suspected infection
DOI: 10.1590/S1679-45082011AO2020
ABSTRACT
Objective:
To compare efficacy and safety of vancomycin versus teicoplanin in patients with proven or suspected infection.
Methods:
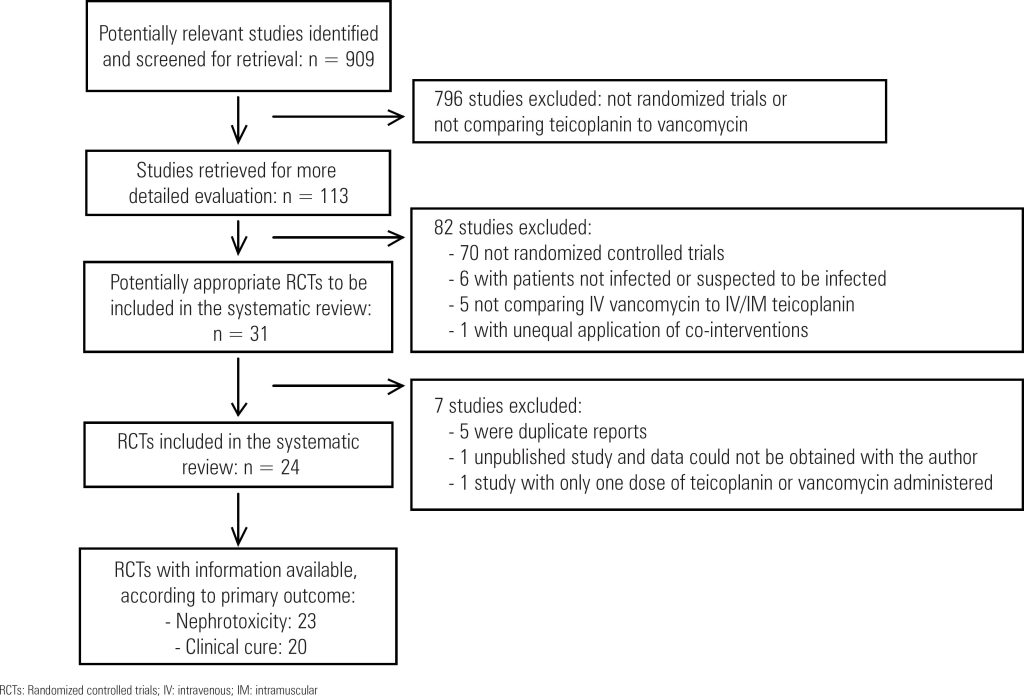
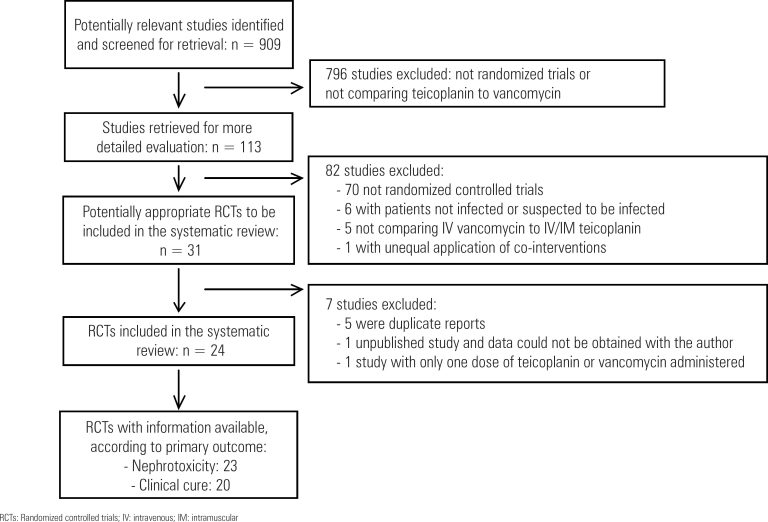
Data Sources: Cochrane Renal Group’s Specialized Register, CENTRAL, MEDLINE, EMBASE, nephrology textbooks and review articles. Inclusion criteria: Randomized controlled trials in any language comparing teicoplanin to vancomycin for patients with proven or suspected infection. Data extraction: Two authors independently evaluated methodological quality and extracted data. Study investigators were contacted for unpublished information. A random effect model was used to estimate the pooled risk ratio (RR) with 95% confidence interval (CI).
Results:
A total of 24 studies (2,610 patients) were included. The drugs had similar rates of clinical cure (RR: 1.03; 95%CI: 0.98-1.08), microbiological cure (RR: 0.98; 95%CI: 0.93-1.03) and mortality (RR: 1.02; 95%CI: 0.79-1.30). Teicoplanin had lower rates of skin rash (RR: 0.57; 95%CI: 0.35-0.92), red man syndrome (RR: 0.21; 95%CI: 0.08-0.59) and total adverse events (RR: 0.73; 95%CI: 0.53-1.00). Teicoplanin reduced the risk of nephrotoxicity (RR: 0.66; 95%CI: 0.48-0.90). This effect was consistent for patients receiving aminoglycosides (RR: 0.51; 95%CI: 0.30-0.88) or having vancomycin doses corrected by serum levels (RR: 0.22; 95%CI: 0.10-0.52). There were no cases of acute kidney injury needing dialysis.
Limitations:
Studies lacked a standardized definition for nephrotoxicity.
Conclusions:
Teicoplanin and vancomycin are equally effective; however the incidence of nephrotoxicity and other adverse events was lower with teicoplanin. It may be reasonable to consider teicoplanin for patients at higher risk for acute kidney injury.
2,232