einstein (São Paulo). 23/abr/2026;24:eAO1908.
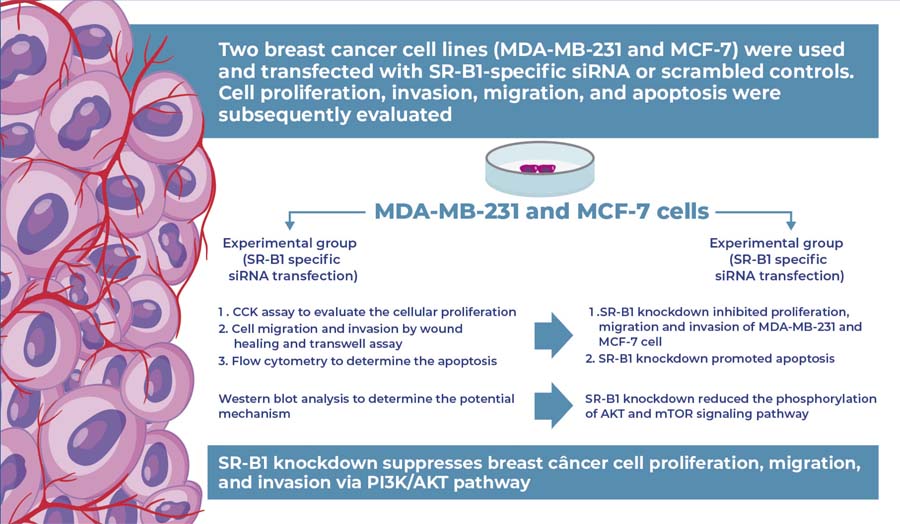
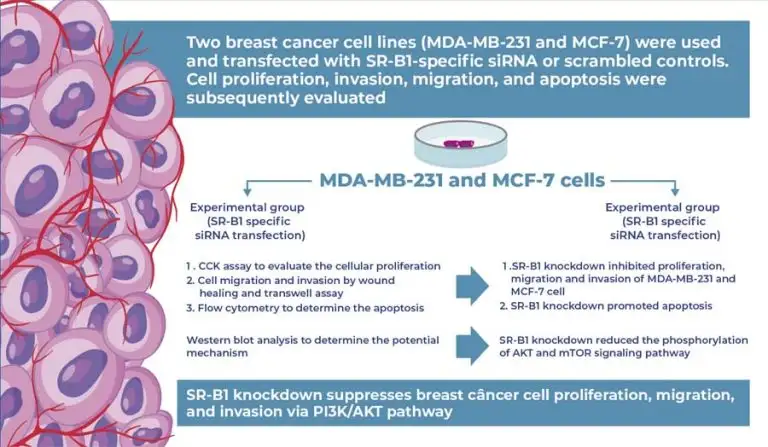
SR-B1 knockdown suppresses breast cancer cell proliferation, migration, and invasion via the PI3K/AKT pathway
DOI: 10.31744/einstein_journal/2026AO1908
Highlights
■ SR-B1 knockdown inhibits breast cancer cell proliferation and migration.
■ Silencing SR-B1 promotes apoptosis in breast cancer cells.
■ PI3K/AKT pathway activity decreases after SR-B1 knockdown.
■ Cyclin D1 and P70 are downregulated following SR-B1 silencing.
ABSTRACT
Background:
The role of scavenger receptor class B type 1 (SR-B1) in breast cancer remains largely unknown.
Objective:
This study aimed to investigate the effects of SR-B1 on breast cancer cell proliferation, migration, and invasion and to elucidate the underlying mechanisms.
Methods:
Two breast cancer cell lines, MDA-MB-231 and MCF-7, were used in this study. Cells were transfected with SR-B1-specific siRNA (si-SR-B1), whereas cells transfected with scrambled sequences served as controls. Cell proliferation was assessed using CCK-8 and colony formation assays. Cell migration and invasion were evaluated using wound healing and Transwell assays, respectively. Apoptosis was analyzed using flow cytometry. Western blot analysis was performed to examine activation of the PI3K/AKT signaling pathway following SR-B1 knockdown.
Results:
Knockdown of SR-B1 significantly inhibited the proliferation, migration, and invasiveness of MDA-MB-231 and MCF-7 cells (all p<0.05). Moreover, SR-B1 knockdown promoted apoptosis in these cells. Western blot analysis revealed that the phosphorylation levels of AKT and mTOR were markedly decreased in the si-SR-B1 group compared to those in the controls. Additionally, the expression of downstream targets, including cyclin D1 and P70, was downregulated by SR-B1 silencing.
Conclusion:
SR-B1 contributes to the enhanced proliferation and migration of breast cancer cells, likely through activation of the PI3K/AKT signaling pathway.
[…]
29